Photosynthesis
Utilization of solar energy in oxygenic photosynthesis
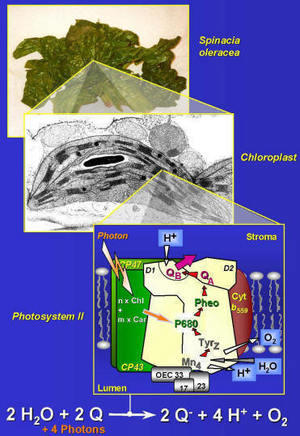
In plants and cyanobacteria, light-driven cleavage of water and release of molecular oxygen takes place at a relatively large protein complex, the Photosystem II (PS II), which is embedded in a lipid bilayer membrane. Because nearly all of the atmosphere's dioxygen has been produced and is continuously replenished by PS II water-splitting, the processes taking place at PS II are of fundamental importance for the earth's atmosphere and biosphere. A complete understanding of the functional and regulatory mechanisms of oxygenic photosynthesis could stimulate technical use of solar energy and lead to new perspectives in biotechnology.
|
|
The Photosystem II represents a highly complex nanocatalyst. Four functional areas can be discriminated:
1. Excitation energy transfer between ca. 200 'antenna molecules' (chlorophylls, carotenoids, ..)
2. Ultra-fast intramolecular electron transfer
3. Stabile reduction of mobile electron acceptors (quinones)
4. Usage of water as a substrate (source of electrons and protons) catalyzed by a protein-bound metal complex, the tetra-manganese complex of PS II |
Because these reaction are 'triggered' by light pulses and many intrinsic 'sensors' can be used, photosystems are well-suited for studying several aspects of the physics of biological macromolecules.
During the recent years, we worked on questions concerning all four functional areas of photosystems. Keywords are: exciton equilibration in PS II antenna, assembly of pigment-protein complexes, function of chlorophylls absorbing in the long-wavelength region, influence of electric fields on energetics and kinetics of primary charge separation, substrate and inhibitor binding at the acceptor site, pH-induced formation of macro-aggregates, mechanisms protecting against photooxidative damage.
|
|
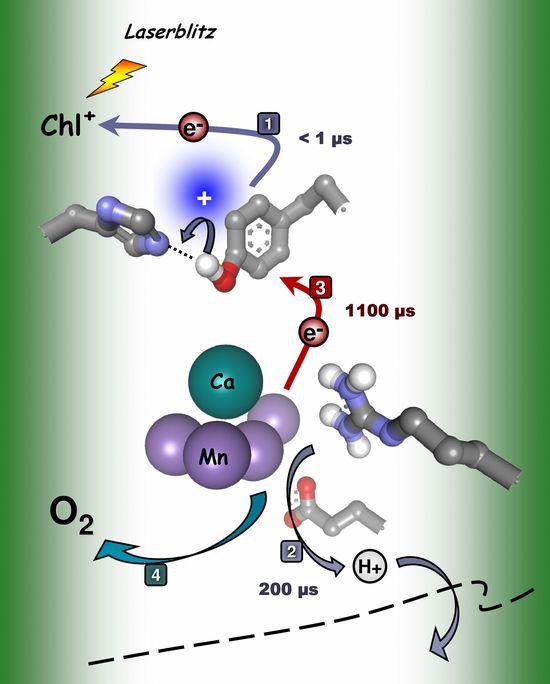
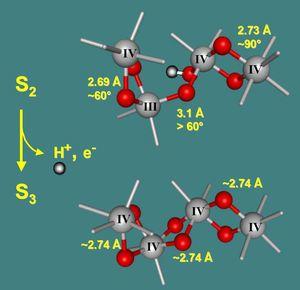
At present, work in our group focuses on water oxidation by the PS II manganese complex. Our efforts aim towards elucidation of the mechanism of water cleavage (or make significant contributions) by examination of structural changes of the manganese complex during its functional cycle with X-ray absorption spectroscopy. Recent progress has facilitated tracking of the manganese complex in its reaction cycle by time-resolved X-ray absorption spectroscopy after Laser-pulse excitation. The figure below summarizes central results of the time-resolved experiments. (Haumann, Liebisch, Müller, Grabolle, Barra, and Dau (2005) SCIENCE 310, 1019-1021.) |