Time-resolved UV/Vis spectroscopy
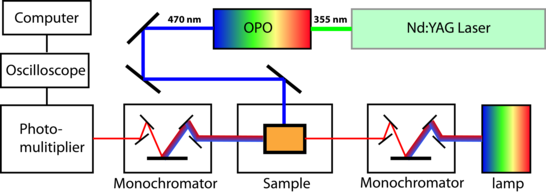
An UV/Vis spectrometer detects absorption of a sample in the UV/Vis region. Polychromatic light is emitted by a lamp. The attached monochromator selects a specific wavelength that is transferred to the sample chamber. The light that passes the sample (may be reduced in intensity) is absorbed by the detector, a photomultiplier. An oscilloscope records the voltage and transferred the signal to a computer.
To detect time-dependent absorption changes (kinetics) a flash photolysis is used. The setup is similar to a UV/Vis spectrometer. In addition a pulsed laser is attached to activate light sensitive proteins. The second monochromator after the sample chamber prevents scattered light from the laser to hit the detector (figure 1).
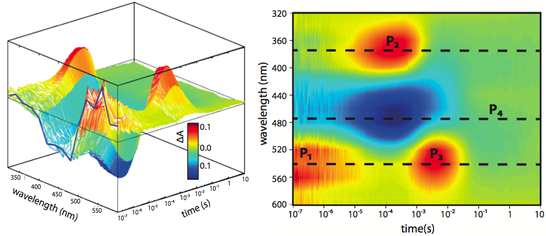
After light activation retinal proteins like bacteriorhodopsin (bR) undergo a photocycle (view projects/retinal proteins). Not all proteins are activated by the laser falsh, therefore the intensity of the absorption changes are relatively small. Difference spectroscopy allows us to detect these changes. For this the ground state is subtracted as a background which means that the resulting kinetic shows only the absorption changes related to light activation. The kinetic are detected for each wavelength separately and add up to a 3D spectrum composed of time, wavelength and absorption changes (figure 2).