Cytochrome c oxidase
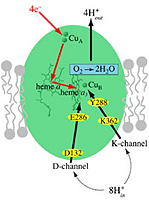
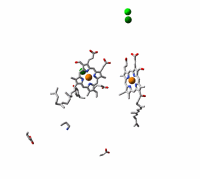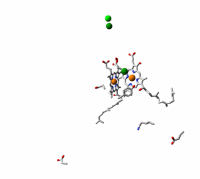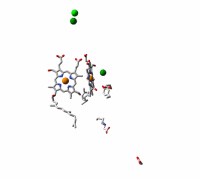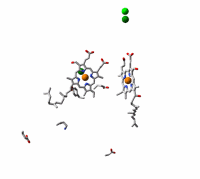
Cytochrome c oxidase, the terminal enzyme in the mitochondrial respiratory chain, performs the four-electron reduction of molecular oxygen to water and the translocation of four protons. The net movement of these charges contributes to an electrochemical gradient across the membrane. This free energy gradient is used e.g. by the ATP synthase to make the primary fuel for cellular processes. We have examined the kinetics of changes in the protein and cofactors when carbon monoxide, an inhibitor of the active site, is photolyzed from fully reduced CO-bound cytochrome c oxidase. We aim at elucidating the coordination and sequence of electron transfer, proton pumping, and oxygen chemistry in the enzyme.